Clearing expired medications isn’t just about tidying up shelves. It’s a safety issue that can prevent harm, avoid legal trouble, and stop thousands of dollars from being wasted. If you’re handling meds in a pharmacy, clinic, or even a home care setting, getting this right matters. Too many people make the mistake of guessing expiration dates from lot numbers - and that’s dangerous. The truth is simple: expiration date is the only thing you should trust. Lot numbers? They’re for tracing, not timing.
Why You Can’t Guess Expiration from Lot Numbers
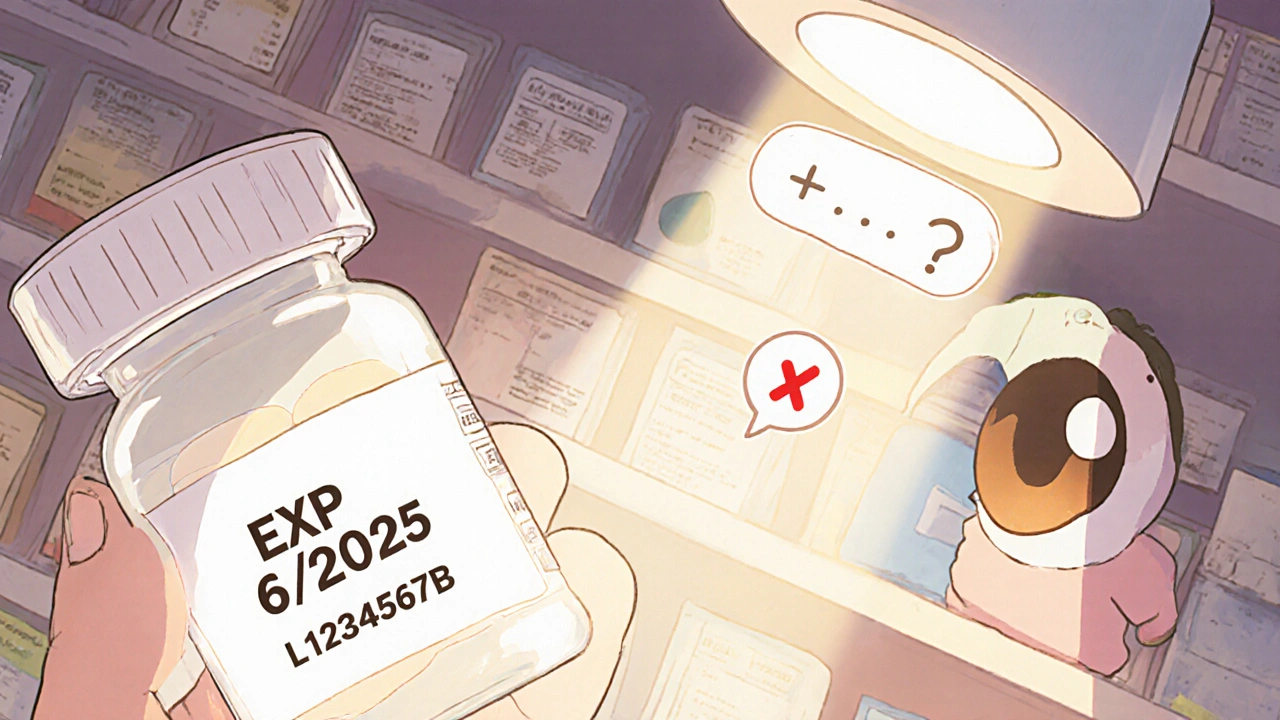
Lot numbers are not expiration codes. They’re batch identifiers. Think of them like a car’s VIN - they tell you which factory made it, when it rolled off the line, and which parts were used. But they don’t say when it stops working. Manufacturers use different formats. Pfizer might write 230515A - that’s May 15, 2023, and a production line code. Merck could use MK22B047, where 22 means 2022. But here’s the catch: even if you spot a date in the lot number, you still don’t know how long the drug lasts. A pill might be good for 2 years. Another for 5. It depends on the formulation, storage, and manufacturer. The FDA doesn’t let companies hide expiration dates inside lot numbers. They require the EXP date to be printed plainly on the package - usually in MM/YYYY or DD/MM/YYYY format. A 2023 study found that 38% of international meds use day-month-year format. If you’re used to month-year, you might think a pill expires in June 2025 when it actually expires in December 2025. That’s a 6-month mistake. And mistakes like that lead to either dangerous overdoses of old meds… or throwing out perfectly good ones.The Only Reliable Way to Check Expiration
Look at the package. Not the bottle label. Not the box. Not the lot number. The EXP date. It’s required by law. If it’s faded, torn, or missing? Don’t guess. Don’t assume. Don’t risk it. In 2024, Harvard Medical School tested 1,200 pharmacy inventory checks. When staff relied on EXP dates alone, they got 98.6% accuracy. When they tried to reverse-engineer expiration from lot numbers? Accuracy dropped to 41%. That’s not a typo. Four out of ten times, they got it wrong. Here’s what to do:- Find the EXP label. It’s usually on the side or bottom of the bottle, blister pack, or box.
- If it’s EXP 06/2025, the drug expires at the end of June 2025.
- If it’s EXP 15/06/2025, it expires on June 15, 2025.
- If you can’t read it, take a photo with good lighting and ask a pharmacist. Or call the manufacturer.
How to Check for Recalls Using Lot Numbers
Once you’ve confirmed the expiration date, you need to check if that exact batch has been recalled. A drug can be expired and safe - or it can be expired and dangerous because of contamination, mislabeling, or manufacturing flaws. The FDA maintains a public database called Recalls, Market Withdrawals & Safety Alerts. You can search it by drug name, lot number, or manufacturer. But here’s the trick: you need the exact lot number from the package. No approximations. No partial matches. For example, if your bottle says L1234567B, you type L1234567B into the search bar - not L1234567. One character off, and you miss a recall. Pharmacies in the U.S. are required to check this database before disposing of any expired meds. In 2021, the FDA documented 217 recall incidents where expired meds stayed in stock because staff didn’t cross-check lot numbers. Some of those meds were linked to serious infections and overdoses. You can also call the manufacturer directly. Most have a toll-free number on the packaging. If you’re unsure, ask the pharmacy’s compliance officer. They’re trained to handle this.
What Happens If You Skip the Check?
Ignoring lot numbers and recalls isn’t just sloppy - it’s risky. In 2023, a chain pharmacy in Ohio gave out 1,200 doses of expired antibiotics. The lot number matched one that had been recalled for bacterial contamination. Patients got sick. One ended up in the ICU. The pharmacy paid $2.3 million in fines and settlements. Even if nothing bad happens, you’re still breaking the law. The DEA requires all controlled substances to be tracked and documented when disposed. The FDA requires all meds to be cleared properly. If you’re audited and can’t show you checked recalls and expiration dates? You could lose your license. And financially? It’s expensive. A 2024 Gartner report found that pharmacies waste $412 million a year on expired meds - partly because they misread lot numbers and threw out good drugs, or kept bad ones too long.How to Set Up a Safe Clearance Process
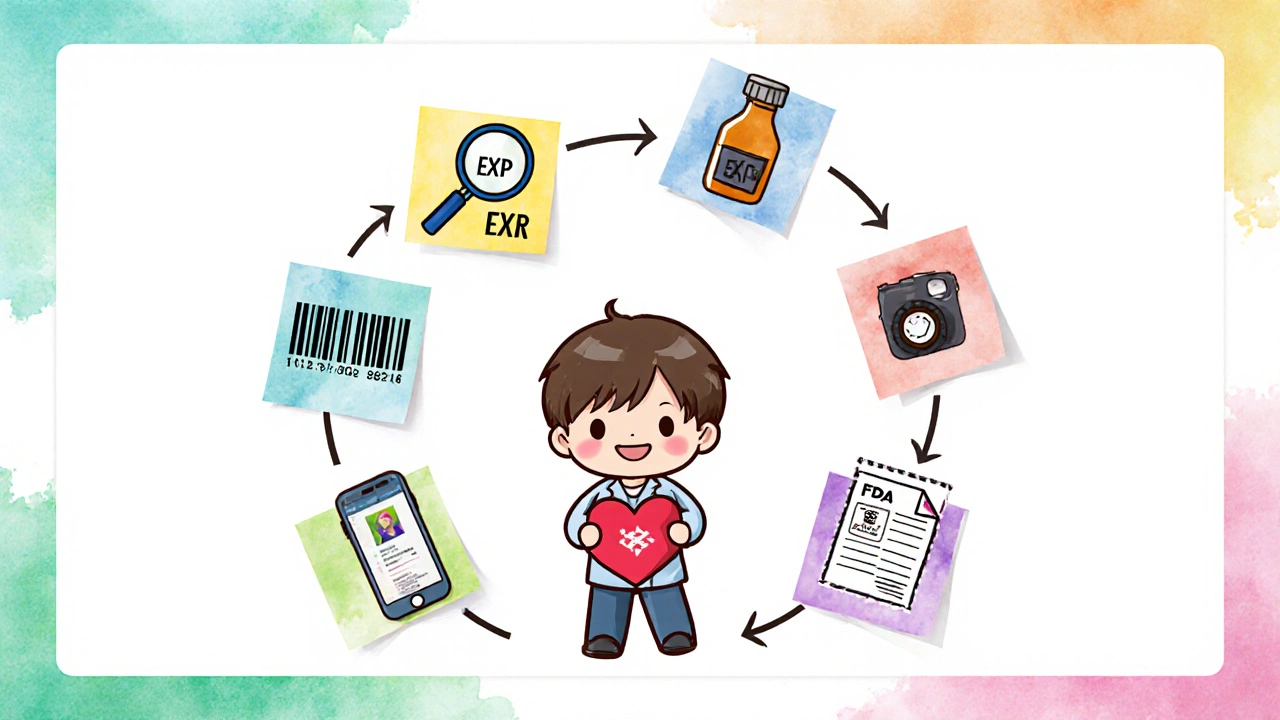
If you’re managing meds regularly, you need a system. Here’s a simple 7-step process used by top pharmacies:- Isolate any meds with EXP dates within 60 days. Don’t wait until they’re expired.
- Scan the lot number into your inventory system. If you don’t have one, write it down.
- Go to the FDA’s recall database and search that lot number.
- Call the manufacturer’s customer service line and confirm there’s no active recall.
- Take a timestamped photo of the bottle with the EXP date and lot number visible.
- For controlled substances, fill out FDA Form 3639 before disposal.
- Keep all records - photos, logs, emails - for at least two years.
Tools That Make This Easier
You don’t have to do this manually. Systems like MedKeeper, IFS Inventory, and GS1-compliant scanners automate the process. They scan the lot number and EXP date at the same time, cross-check the FDA database, and flag anything suspicious. The FDA approved an AI scanner in April 2024 that reads EXP dates from damaged or blurry labels with 99.2% accuracy. That’s huge. Because 31% of medication labels get smudged, bent, or faded during shipping and storage. Even if you’re not using fancy tech, you can still improve. Use a bright LED lamp - 500+ lux - when reading labels. Poor lighting causes 1 in 5 misreads. And update your manufacturer contact list every quarter. Companies change phone numbers. Websites get redesigned. Don’t rely on old info.What’s Changing in 2025 and Beyond
By November 2025, the FDA will require all pharmacies to use electronic lot tracking. That means no more paper logs. No more handwritten notes. Everything must be digital and traceable. The industry is moving toward standardized lot numbers through the SNI (Standardized Numerical Identification) initiative. But here’s the key: expiration dates will still be separate. The FDA made that clear in 2023. They won’t merge them. So even when lot numbers get simpler, you’ll still need to read the EXP label. By 2027, experts predict over 89% of manufacturers will use GS1 barcode standards. That means scanning a pill bottle will give you the drug name, lot number, and expiration date - all in one scan. That’s the future. But until then, you still have to do the work.Final Rule: Trust the Label, Not the Code
Lot numbers are for tracing. Expiration dates are for safety. Recalls are for protection. You need all three. If you only remember one thing: never calculate expiration from a lot number. It’s not just wrong - it’s illegal. And it puts lives at risk. When in doubt:- Look for the EXP date.
- Search the FDA recall database with the exact lot number.
- When in doubt, call the manufacturer.
- Document everything.
Can I use the lot number to find out when my medicine expires?
No. Lot numbers are batch identifiers, not expiration codes. Manufacturers use different formats, and there’s no public database that links lot numbers to expiration dates. The only reliable way to know when a medicine expires is to read the printed "EXP" date on the package. Guessing from the lot number can lead to dangerous mistakes.
How do I check if my medication has been recalled?
Go to the FDA’s official Recalls, Market Withdrawals & Safety Alerts page and enter the exact lot number from your medication’s packaging. You can also call the manufacturer’s customer service line - the number is printed on the box or bottle. Never rely on memory, partial numbers, or online forums. Only official sources are trustworthy.
What if the expiration date on my pill bottle is faded or missing?
Do not use the medication. If the EXP date is unreadable, contact the pharmacy that dispensed it or the manufacturer directly. Take a photo with good lighting and ask for help. In a professional setting, such medications must be quarantined and reported as damaged inventory. Never assume an expiration date - even if you think you remember it.
Why do some medications have different expiration formats like DD/MM/YYYY vs MM/YYYY?
The FDA requires expiration dates to be in a clear calendar format, but doesn’t mandate a single style. Many international manufacturers use DD/MM/YYYY, while U.S. labels typically use MM/YYYY. This can cause confusion, especially for staff unfamiliar with the format. Always double-check the date format before assuming when a drug expires. A date like "06/2025" could mean June 2025 or June 6, 2025 - context matters.
Is it safe to use expired medicine if it looks fine?
No. Even if a pill looks unchanged, its potency can drop over time, or it can degrade into harmful substances. Some antibiotics, for example, become toxic after expiration. The FDA states that expired medications should not be used unless under specific emergency guidance - which rarely applies to routine use. Always dispose of expired meds properly.
Do I need to document expired medication disposal?
Yes. For all medications, especially controlled substances, you must document the disposal. This includes the drug name, lot number, expiration date, quantity, date of disposal, and your initials. For controlled substances, you must complete FDA Form 3639. Records must be kept for at least two years. Failure to document can result in fines or loss of licensure.





Savakrit Singh
November 27, 2025 AT 11:39OMG this is LIFE-SAVING info!! 🙌 I work in a clinic in Delhi and we used to guess expiration dates from lot numbers because the labels were always smudged... until last month when a patient had a reaction. 😱 Now we scan everything, take photos, and call manufacturers. Thank you for this! 📸💊 #SafetyFirst
Leo Adi
November 28, 2025 AT 08:28Interesting. In Indonesia, we often rely on pharmacists to interpret lot numbers because the packaging is so inconsistent. But I see your point - if the FDA requires a clear EXP label, then we’re all just guessing when we don’t look at it. Still, I wonder how many people even know what 'EXP' stands for?
Mira Adam
November 29, 2025 AT 04:54Let me get this straight - people are still using LOT NUMBERS to guess expiration dates? Like, in 2025? After all the lawsuits, all the deaths, all the FDA warnings? This isn’t ignorance. It’s negligence. And if you’re doing this, you’re not just untrained - you’re dangerous. Stop making excuses. Read the label. Or get out of healthcare.
Miriam Lohrum
November 29, 2025 AT 21:56There’s something deeply human about our need to decode things - even when we’re told not to. Lot numbers feel like secrets we’re meant to crack. But expiration dates? They’re quiet. They don’t reward curiosity. They just exist. And maybe that’s why we ignore them. Not because we’re stupid - but because we’re searching for meaning where there’s only regulation.
archana das
December 1, 2025 AT 03:25My aunt in Mumbai used to keep her blood pressure pills for years because 'they still look fine'. I showed her this post. She cried. Then she threw them all out. 💔 Medicine isn’t just chemicals - it’s trust. And we break that trust every time we guess. Thank you for speaking up.
Emma Dovener
December 2, 2025 AT 13:07Just a quick note: if you’re in a home care setting and the EXP date is faded, call the pharmacy that dispensed it. They’re legally required to keep records for 7 years. Most have digital archives. Don’t waste time with Google or forums - go straight to the source. It’s faster, safer, and you’ll feel better about it.
Sue Haskett
December 4, 2025 AT 04:41PLEASE. PLEASE. PLEASE. Don’t assume. Don’t guess. Don’t ‘hope it’s fine’. The FDA database is free. The manufacturer’s phone number is printed on the box. Taking 3 minutes to verify could save a life - or keep you from losing your job, your license, your freedom. This isn’t optional. It’s non-negotiable. Write it on your mirror. Tell your team. Post it in the break room. Do it.
Jauregui Goudy
December 5, 2025 AT 01:59Bro, I used to be the guy who’d say ‘eh, it’s probably fine’ - until I saw a guy in the ER with a kidney infection from expired amoxicillin. That was my fault. I didn’t check the lot. I didn’t call. I just tossed it in the bin. Now I do the 7-step like clockwork. It takes 22 minutes. And I sleep better. If you’re lazy - you’re not just risking yourself. You’re risking someone’s kid. Wake up.
Rhiana Grob
December 6, 2025 AT 14:20I appreciate how this post balances urgency with clarity. Too many safety guides feel like lectures. This one says: here’s what’s true, here’s what’s at stake, here’s how to fix it - without shame. That’s rare. And necessary. Thank you for making compliance feel like care, not control.
Jonah Thunderbolt
December 6, 2025 AT 23:41Wow. Just... wow. I’ve been doing this wrong for 12 years. And now I’m supposed to believe that some guy with a blog and a 2024 Harvard study is the reason I’m not in jail? 🤡 I mean, I get it - but I also get that this is just another corporate compliance theater. Next thing you know, they’ll make us scan our socks for recalls. 😒
Rebecca Price
December 8, 2025 AT 15:54Jonah - I hear your sarcasm. And I get it. Compliance feels like bureaucracy. But let me ask you this: if your mother took a pill that degraded into a toxin because someone skipped the lot check… would you still call it ‘theater’? Or would you want someone to have done the work so she didn’t have to die? This isn’t about rules. It’s about who gets to live. And that’s worth every minute.